Iron standard solution traceable to SRM from NIST Fe(NO₃)₃ in HNO₃ 0.5 mol/l 1000 mg/l Fe Certipur® | Sigma-Aldrich
プラチナ万年筆 PLATINUM オ・レーヌ OLEeNU / プロユース PRO-USE 製図用 ペンシル 耐芯構造 0.3mm / 0.5mm / 0.7mm MSD-1000 MOL-1000 :yahoo-pla-mol-1000-1:You STYLE - 通販 - Yahoo!ショッピング

Moles and Solutions SPECIFICATIONS Moles and solutions Calculate the amount of substance in moles using solution volume and concentration. - ppt download

SOLVED: Useful Information mole solute L solution M M,Vi = M-Vz mole particles mol AT; 1.86 ATb 0.51 kg solvent kg 1000 mL= 1L 1000 g = 1kg Question 9 pts Astock
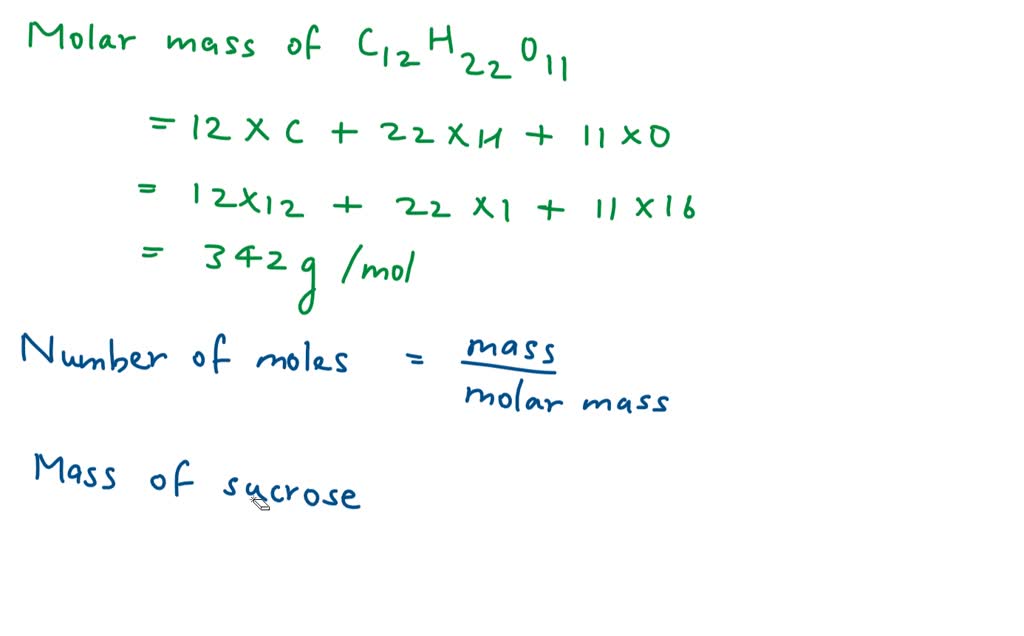
SOLVED: How many moles are equivalent to 342 Kg of sucrose (C12 H22O11)?(a) 10 mol b) 1 mol c) 100 mol d) 1000 mol