![SOLVED: Which is the CORRECT Ksp expression for Fe2(CO3)3? Ksp 0 [Fe]? [CO3]' [Fe3+j? [CO32-13 [Fest] [8O321 / [Fez(COs):] [Fe3+j? [CO32-1 / [Fez(CO3)3] [Fe]? [CO3]? [Fez(CO3)3] [Fes+] [CO3?-] SOLVED: Which is the CORRECT Ksp expression for Fe2(CO3)3? Ksp 0 [Fe]? [CO3]' [Fe3+j? [CO32-13 [Fest] [8O321 / [Fez(COs):] [Fe3+j? [CO32-1 / [Fez(CO3)3] [Fe]? [CO3]? [Fez(CO3)3] [Fes+] [CO3?-]](https://cdn.numerade.com/ask_images/6b9de619ef914826b94338cd7f5d81af.jpg)
SOLVED: Which is the CORRECT Ksp expression for Fe2(CO3)3? Ksp 0 [Fe]? [CO3]' [Fe3+j? [CO32-13 [Fest] [8O321 / [Fez(COs):] [Fe3+j? [CO32-1 / [Fez(CO3)3] [Fe]? [CO3]? [Fez(CO3)3] [Fes+] [CO3?-]
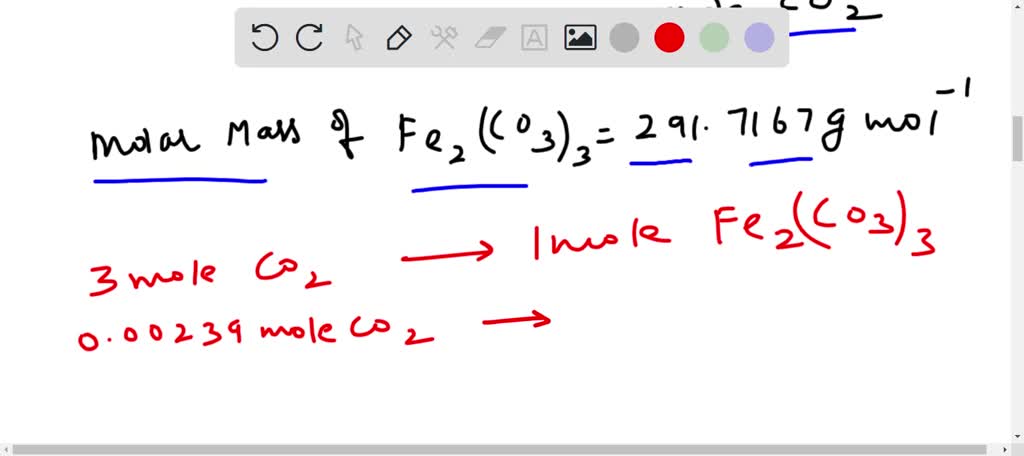
SOLVED: How many grams of iron(III) carbonate decompose to give 53.6 mL of carbon dioxide gas at STP? Fe2(CO3)3??Fe2O3(s)+3CO2(g)
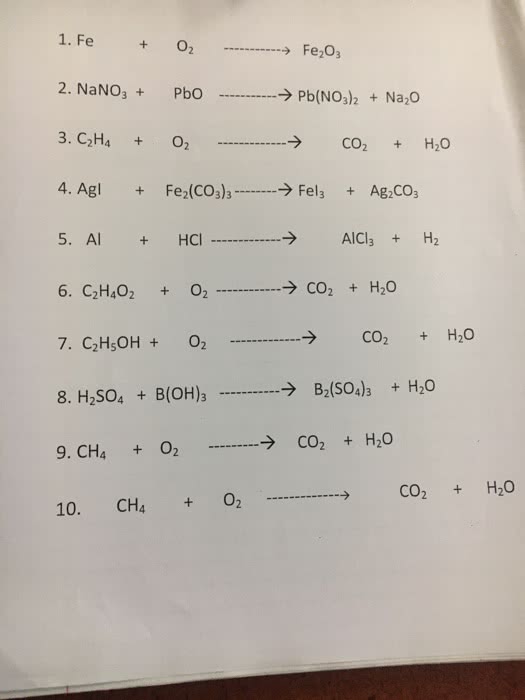
OneClass: 2. NaNO3+ PbO â†'Pb(NOJ2 + Na20 3. C2H4 + 02 4. Agl + Fe2(CO3)3 â†'Fels + Ag2co, 5. Al + HC...







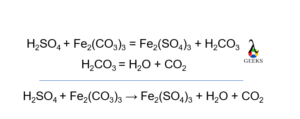
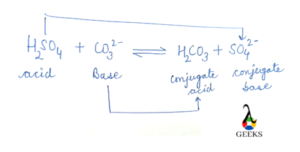

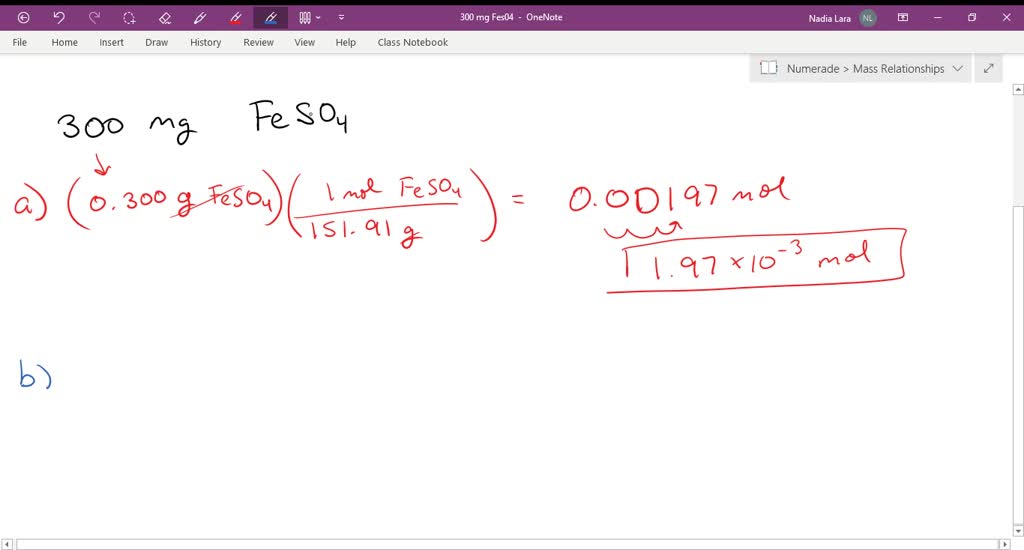

_how-to-write-the-name-for-fe2co33.jpg)



