
Calculate Ecell and Δ G for the following at 28^o C Mg(s) + Sn^2 + (0.025M)→ Mg^2 + (0.06M) + Sn(s) E^0cell = 2.23 VIs the reaction spontaneous?

Three reactions involving H2PO4^- are given below:(i) H3PO4 + H2O→H3O^+ + H2PO4^- (ii) H2PO4^- + H2O→HPO4^2 - + H3O^+ (iii) H2PO4^- + OH^-→H3PO4 + O^2 - In which of the above reactions,

Influence of the composition and concentration of the nutrient solution on plants grown in sand cultures . n> <Z CD n (i I. n 0 c 03 > CI -p COO ?< ?

Influence of the composition and concentration of the nutrient solution on plants grown in sand cultures . PLATE 15 Treatment NaNO:!Grams Ca(H2P04)2Grams K,SO,Grams MgSO,Grams TotalGrams Pot ID2 .2 .3 .6 .24 1.34 Pot IID2 .75 .1 .6 .24 1.69 Pot ...

Figure 1 from The crystal structure of mercury(I) dihydrogenphosphate, Hg2(H2PO4)2 | Semantic Scholar
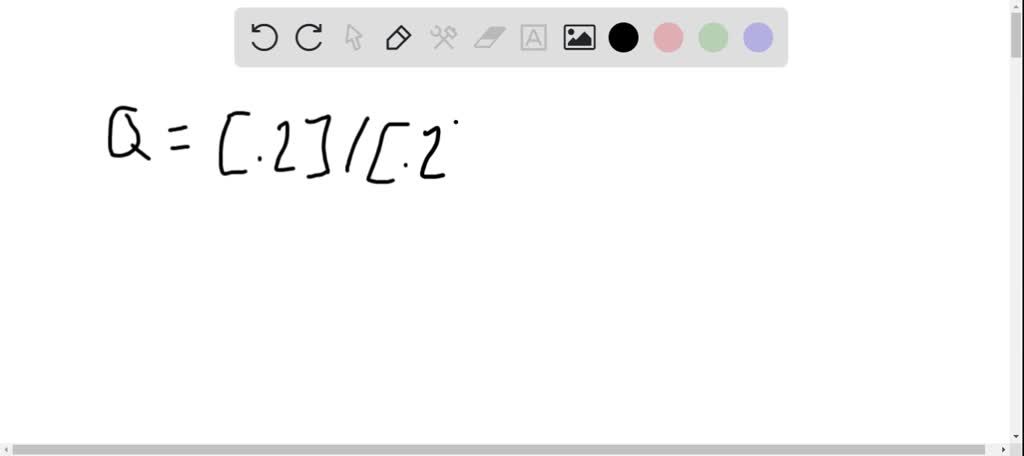
SOLVED: If the initial concentrations of HPO4 and H2C03 areboth equal to 0.100 M, and the initial concentrations of H2P04 andHC03 are both equal to 0.000 M, in which direction will thereaction

L'acido fosforico (acido ortofosforico, H2P04) è una molecola di acido minerale e debole. Formula chimica strutturale e modello molecolare. Foglio di carta in una Immagine e Vettoriale - Alamy
![Preparation and characterization of magnetite-dihydrogen phosphate as a novel catalyst in the synthesis of tetrahydrobenzo[b]pyrans - ScienceDirect Preparation and characterization of magnetite-dihydrogen phosphate as a novel catalyst in the synthesis of tetrahydrobenzo[b]pyrans - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0254058417305084-gr1.jpg)








