Lithium hexamethyldisilazide initiated superfast ring opening polymerization of alpha-amino acid N-carboxyanhydrides | Nature Communications
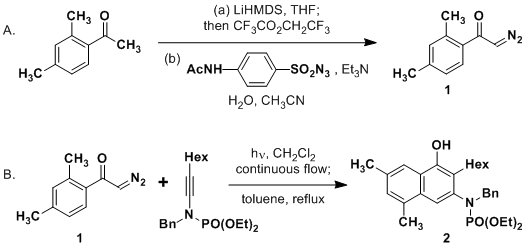
Efficient synthesis of novel N -substituted 2-carboxy-4-quinolones via lithium bis(trimethylsilyl)amide (LiHMDS)-induced in situ cyclocondensation rea ... - RSC Advances (RSC Publishing) DOI:10.1039/C6RA28631C

LiHMDS‐Mediated Deprotonative Coupling of Toluenes with Ketones - Shigeno - Chemistry – A European Journal - Wiley Online Library

LiHMDS: Facile, highly efficient and metal-free transesterification under solvent-free condition - ScienceDirect

LiHMDS: Facile, highly efficient and metal-free transesterification under solvent-free condition - ScienceDirect

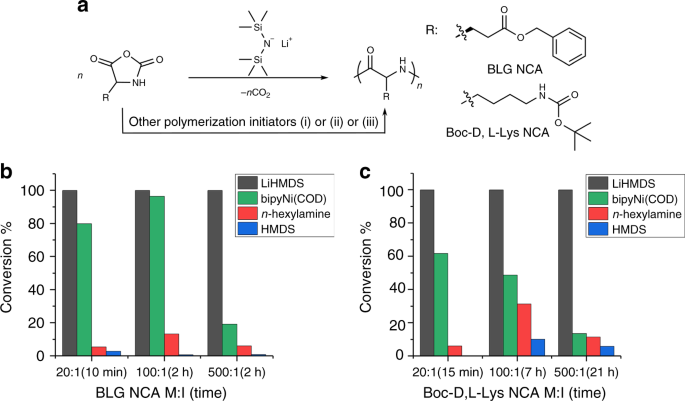
Cyclic Poly(α-peptoid)s by Lithium bis(trimethylsilyl)amide (LiHMDS)-Mediated Ring-Expansion Polymerization: Simple Access to Bioactive Backbones | Journal of the American Chemical Society
Lithium Hexamethyldisilazide-Mediated Enolization of Acylated Oxazolidinones: Solvent, Cosolvent, and Isotope Effects on Competi

Scheme 55. Reagents: i, LiHMDS, THF, –78ºC; ii, oiodobenzaldehyde ;... | Download Scientific Diagram

Scheme 34 Reagents and condition—a (i) LiHMDS, THF, −78 °C; (ii) R 2 CH... | Download Scientific Diagram
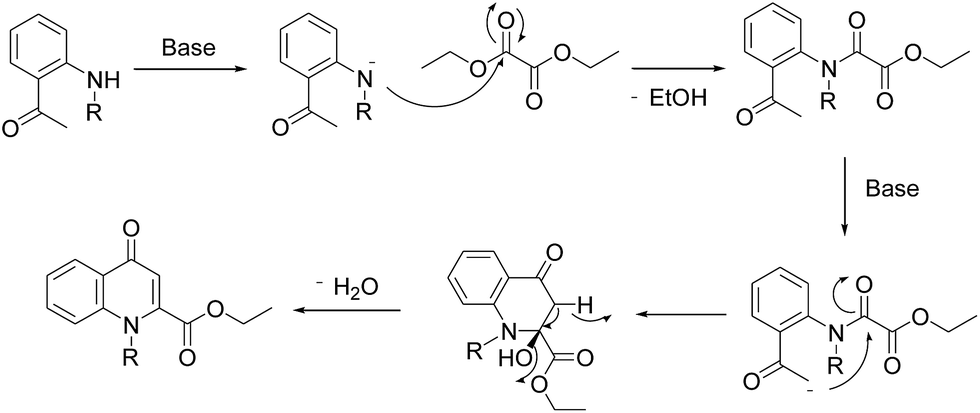
Efficient synthesis of novel N -substituted 2-carboxy-4-quinolones via lithium bis(trimethylsilyl)amide (LiHMDS)-induced in situ cyclocondensation rea ... - RSC Advances (RSC Publishing) DOI:10.1039/C6RA28631C

Cyclic Poly(α-peptoid)s by Lithium bis(trimethylsilyl)amide (LiHMDS)-Mediated Ring-Expansion Polymerization: Simple Access to Bioactive Backbones | Journal of the American Chemical Society

Scheme5.Reagents and conditions: a) 13,LiHMDS, THF, À78 8C; then add... | Download Scientific Diagram

Reaction of Ketones with Lithium Hexamethyldisilazide: Competitive Enolizations and 1,2-Additions | Journal of the American Chemical Society
Lithium Hexamethyldisilazide-Mediated Enolization of Acylated Oxazolidinones: Solvent, Cosolvent, and Isotope Effects on Competi