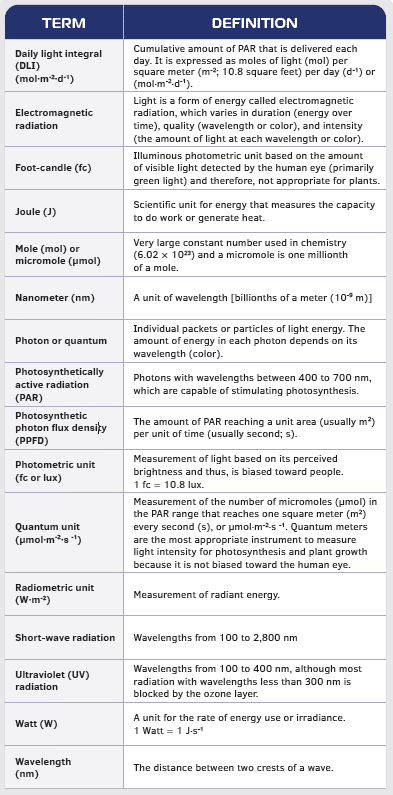
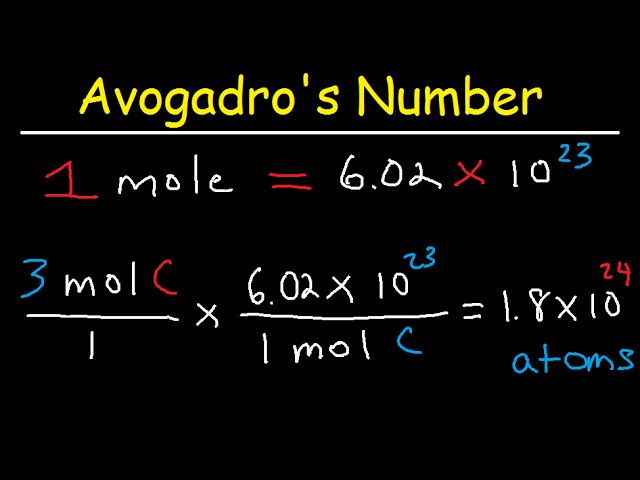SOLVED: Zinc(II) sulfide reacts with oxygen according to the reaction: 2ZnS( s)+3O2(g)→2ZnO(s)+2SO2(g) A reaction mixture initially contains 4.4 mol ZnS and 6.8 mol O2. Once the reaction has occurred as completely as possible,

Redefinition of the Mole in the Revised International System of Units and the Ongoing Importance of Metrology for Accurate Chemical Measurements | Analytical Chemistry

S Mol 250mg Syrup - Sara Remedies | Buy generic medicines at best price from medical and online stores in India - dawaadost.com

Question Video: Determining the Empirical Formula of a Hydrated Salt Given the Mass of the Sample before and after Heating | Nagwa